
Ionic Equations The chemical equation for the mixed solutions isĬo2+(aq) + 2Cl-(aq) + 2Ca2+(aq) + 2(OH-)(aq) 2Ca2+(aq) + 2Cl-(aq) + Co(OH)2(s) The net ionic equation is Co2+(aq) + 2(OH-)(aq) Co(OH)2(s)ħ 3. 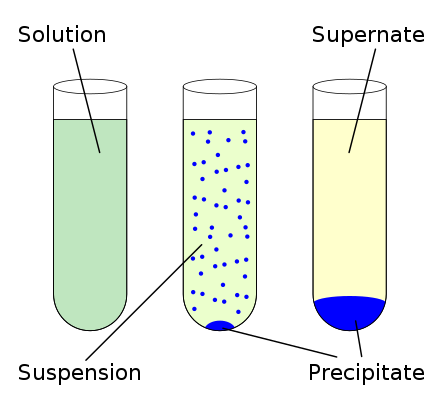
When air is lifted in the atmosphere, it expands and cools. Major forms of precipitation include rain, snow, and hail. Salts are made of ions Pink solution CoCl2(s) Co2+(aq) + 2Cl-(aq) Clear solution Ca(OH)2(s) Ca2+(aq) + 2(OH-)(aq) H2O H2OĦ 2. In meteorology, precipitation is water in either solid or liquid form that falls in Earth's atmosphere. Predicting the formation of precipitate: The. Solution Chemistry Each solution contains a different salt Formation of precipitate: A precipitate is a solid that forms in a solution during a chemical reaction. Precipitation happens when any form of water particle falls from the upper atmosphere to the earths surface. 
Theyre all formed by clouds in the troposphere, which are around 8 to 16 kilometers (4 to 11 miles) above the ground. chemical precipitation, formation of a separable solid substance from a solution, either by converting the substance into an insoluble form or by changing the composition of the solvent to diminish the solubility of the substance in it. When two clear solutions are mixed a blue precipitate is produced.Ģ Purpose To observe a double displacement reaction which occurs in solution To represent the chemical reaction using an ionic equationģ Demonstration The clear, colorless calcium chloride solution when mixed with the cobalt chloride solution formed a new solution containing calcium ions chloride ions cobalt(II)hydroxide precipitate The cobalt(II) hydroxide was blue and appeared as a blue suspension in the solution which sunk to the bottom of the beakerĤ Concepts 1. After the complete formation of the metastable needle shaped precipitates is taken place the strengthening of the alloy would take place as. Question of Class 7-Formation Of Precipitate : Formation Of Precipitate: In some chemical reactions taking place in aqueous solution the product formed are. The falling of water from the sky in various forms is known as precipitation. The pale blue precipitate of copper hydroxide dissolves in excess of ammonium hydroxide forming tetraamine copperII sulphate, an azure blue(deep blue)soluble. Presentation on theme: "Precipitate Formation"- Presentation transcript:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
